What Is the Structure of the Atom?
The structure of the atom is one of the most important foundational topics in chemistry. It explains how atoms—the smallest units of matter—are built and how their internal components determine the properties of elements.
Every substance around us, whether solid, liquid, or gas, is made of atoms. Understanding the structure of the atom helps students explain chemical reactions, bonding, valency, isotopes, and even modern applications like nuclear energy and medical imaging.
In this chapter, we study how scientists gradually discovered the internal structure of the atom through different atomic models. Each model improved upon earlier ideas and brought us closer to the modern atomic theory.
This article explains the structure of the atom step by step, using simple language, clear definitions, worked examples, exam tips, and practice questions—making it ideal for school and competitive exam preparation.
Atom: Basic Definition
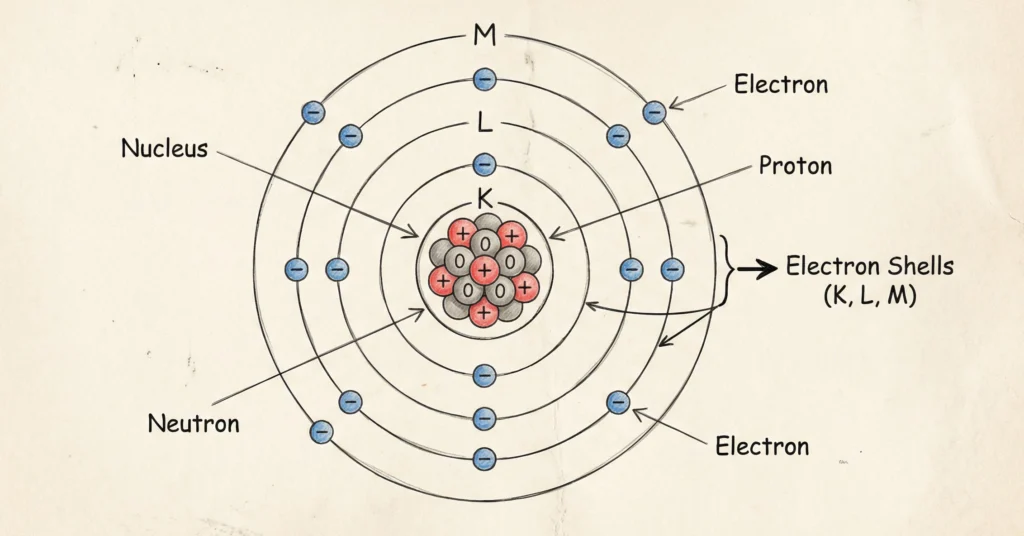
An atom is the basic building block of matter and the smallest unit of an element that can exist independently. An atom is the smallest unit of an element that retains its chemical properties. Every substance around us is made up of atoms, which may combine with one another to form molecules and compounds.
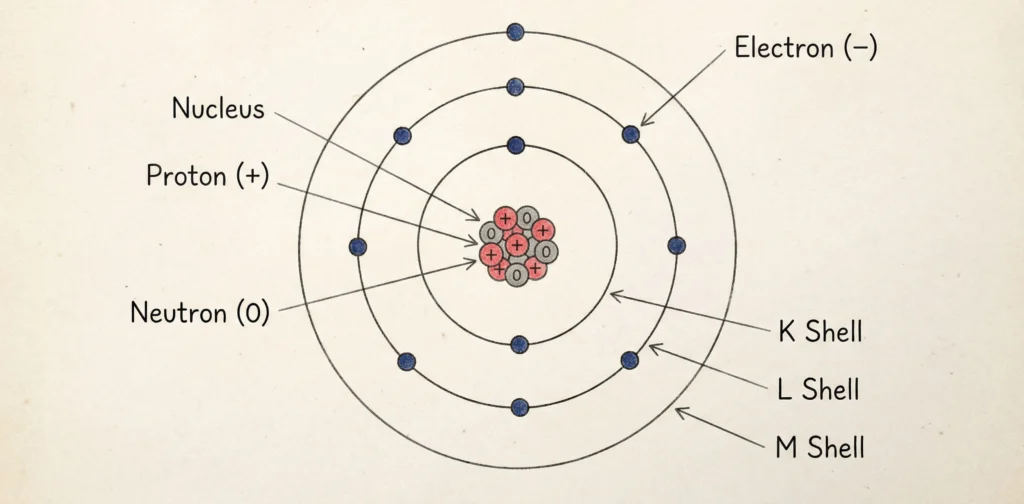
An atom consists of three fundamental subatomic particles: electrons, protons, and neutrons. Protons and neutrons are present in the nucleus at the center of the atom, while electrons revolve around the nucleus in fixed energy levels or shells. The number of protons in an atom determines the identity of the element and is known as the atomic number.
Atoms are electrically neutral because the number of positively charged protons is equal to the number of negatively charged electrons. The structure of the atom plays an important role in determining the physical and chemical properties of elements, making it a fundamental concept in chemistry.
Atoms are made up of three fundamental subatomic particles:
| Particle | Symbol | Charge | Mass (approx.) |
|---|---|---|---|
| Electron | e⁻ | –1 | Negligible |
| Proton | p⁺ | +1 | 1 amu |
| Neutron | n⁰ | 0 | 1 amu |
The structure of the atom refers to the arrangement of these particles inside the atom.
Subatomic Particles Explained
Electrons
- Negatively charged particles
- Revolve around the nucleus
- Determine chemical behavior and bonding
Protons
- Positively charged particles
- Present inside the nucleus
- Decide the atomic number of an element
Neutrons
- Neutral particles
- Add to atomic mass
- Provide stability to the nucleus
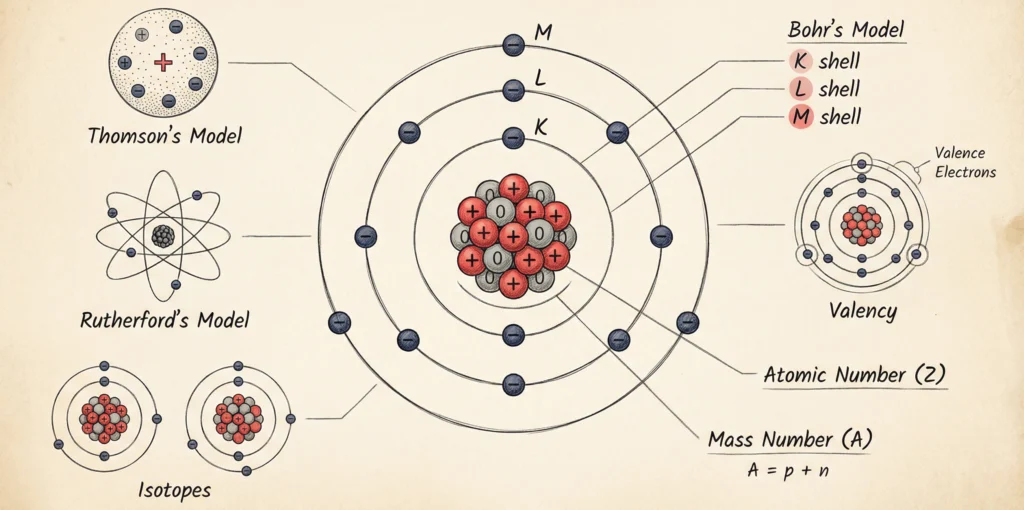
Discovery of Atomic Models
The discovery of atomic models was a gradual process that helped scientists understand the internal structure of the atom. Scientists proposed different models to explain the structure of the atom based on experimental evidence. Each model was developed to describe the arrangement of subatomic particles and to explain the behavior of atoms more accurately.
Initially, atoms were thought to be indivisible. However, the discovery of electrons by J. J. Thomson led to the first atomic model, which suggested that electrons are embedded in a positively charged sphere. Later, Ernest Rutherford’s gold foil experiment revealed the presence of a small, dense, positively charged nucleus, leading to the nuclear model of the atom.
Although Rutherford’s model explained the nucleus, it could not account for atomic stability. This limitation was overcome by Niels Bohr, who proposed that electrons revolve around the nucleus in fixed energy levels. Thus, through continuous experimentation and refinement, atomic models evolved to give a clearer understanding of atomic structure.
Thomson’s Atomic Model
Background
J.J. Thomson discovered electrons in 1897 through cathode ray experiments.
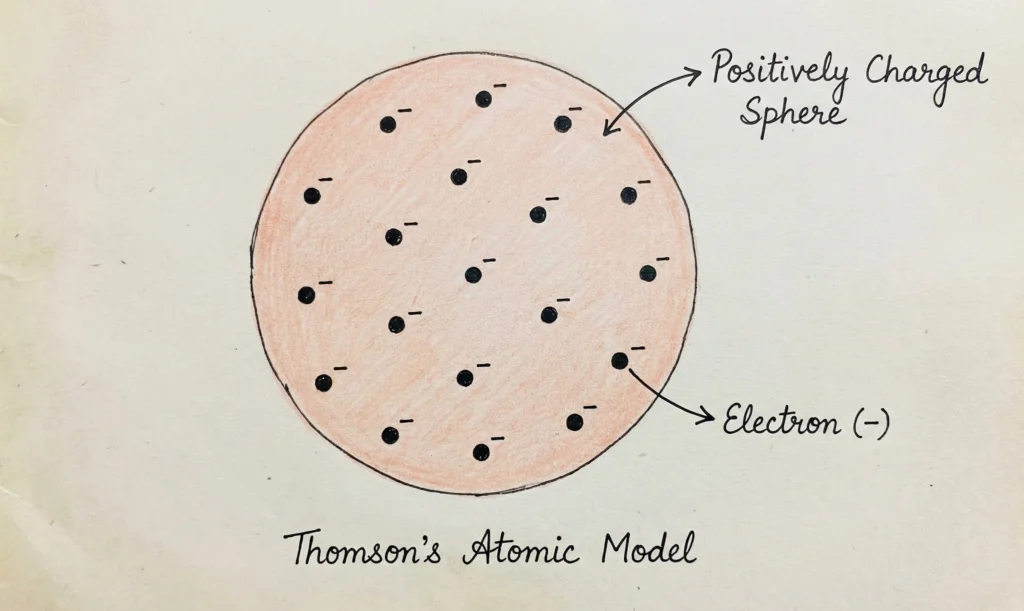
Thomson’s Model (Plum Pudding Model)
According to Thomson:
- The atom is a positively charged sphere
- Negatively charged electrons are embedded in it
- Overall charge of atom is neutral
Merits
- First model to include subatomic particles
- Explained electrical neutrality of atoms
Limitations
- Could not explain atomic stability
- Failed to explain scattering experiment results
Rutherford’s Atomic Model
Gold Foil Experiment
The Gold Foil Experiment was conducted to study the structure of the atom. Ernest Rutherford bombarded thin gold foil with alpha particles. From the observations, he concluded that most of the atom is empty space and that a small, dense, positively charged nucleus is present at the center of the atom.
Observations:
- Very few rebounded
- Most particles passed straight
- Some deflected
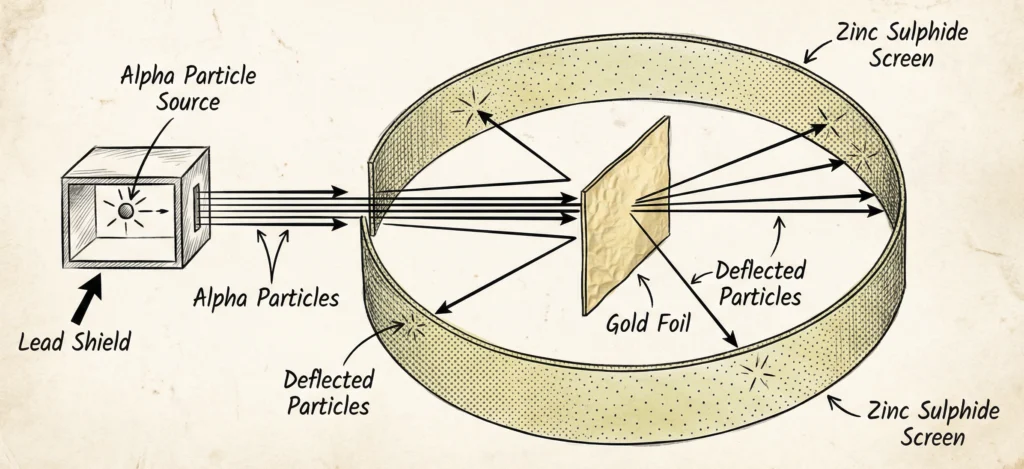
Conclusions
- Atom has a dense, positively charged nucleus
- Most of the atom is empty space
- Electrons revolve around the nucleus
Rutherford’s Atomic Model
- Central nucleus containing protons
- Electrons revolve in circular paths
Limitations
- Could not explain why electrons do not fall into nucleus
- Failed to explain atomic spectra
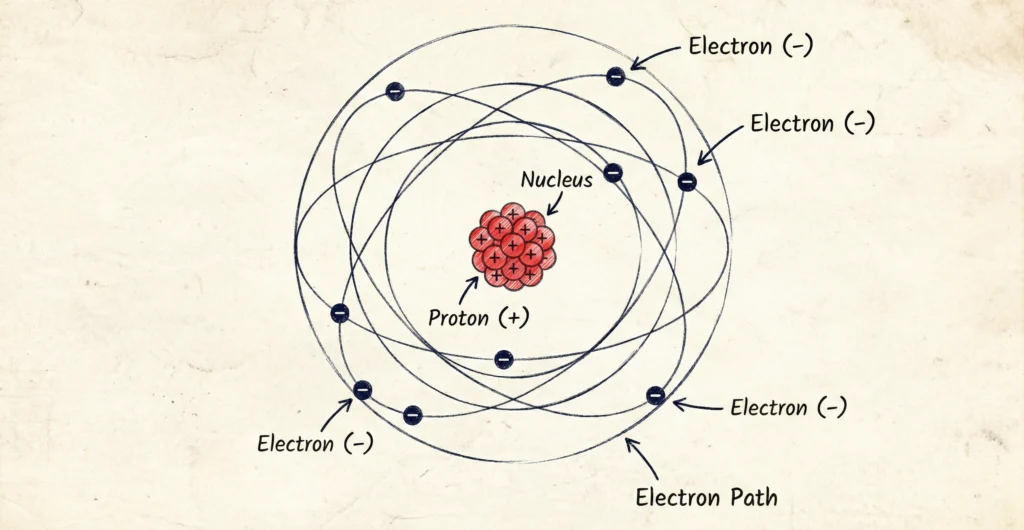
Bohr’s Atomic Model
Need for Bohr’s Model
To overcome Rutherford’s shortcomings, Niels Bohr proposed a new model.
Postulates of Bohr’s Model
- Electrons revolve in fixed circular orbits (energy levels)
- Each orbit has a definite energy
- Energy is emitted or absorbed when electrons jump between orbits
Energy Levels
| Shell | Name | Maximum Electrons |
|---|---|---|
| K | 1st | 2 |
| L | 2nd | 8 |
| M | 3rd | 18 |
| N | 4th | 32 |
Formula:
Maximum electrons = 2n²
(where n = shell number)
Merits
- Explained atomic stability
- Explained hydrogen spectrum
Limitations
- Applicable only to hydrogen-like atoms
- Failed for multi-electron atoms
Valency
Definition
Valency is the combining capacity of an atom. It depends on the number of electrons in the outermost shell. Valency determines how an atom bonds with other atoms to form molecules or compounds. Atoms usually gain, lose, or share electrons to achieve a stable electronic configuration, and the number of electrons involved in this process represents the valency of the element.
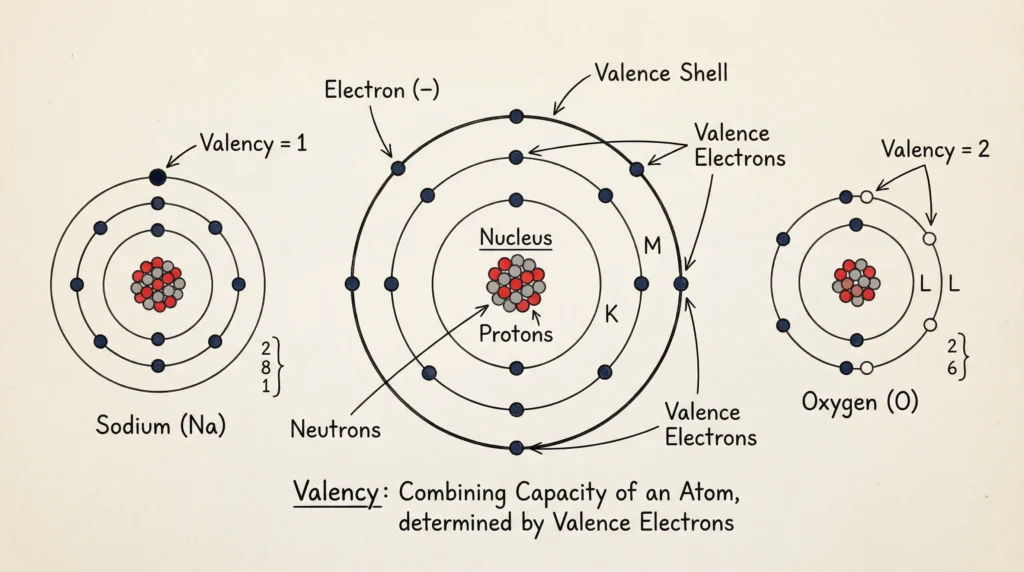
Valency Rules
- If valence electrons ≤ 4 → valency = number of electrons
- If valence electrons > 4 → valency = 8 – number of electrons
Examples
| Element | Valence Electrons | Valency |
|---|---|---|
| Sodium (Na) | 1 | 1 |
| Oxygen (O) | 6 | 2 |
| Neon (Ne) | 8 | 0 |
Atomic Number
Definition
Atomic number (Z) = Number of protons in an atom. It uniquely identifies an element and determines its position in the periodic table. In a neutral atom, the atomic number is also equal to the number of electrons, and it plays a key role in deciding the electronic configuration and chemical properties of the element.
Importance
- Identifies the element
- Determines electronic configuration
- Same for all atoms of an element
Example:
Carbon has 6 protons → Atomic number = 6
Mass Number
Definition
Mass number (A) = Number of protons + neutrons. It represents the total number of nucleons present in the nucleus of an atom. The mass number helps in calculating the number of neutrons in an atom and is useful in distinguishing isotopes of the same element.
Formula
A = Z + n
Where:
- Z = atomic number
- n = number of neutrons
Example
Chlorine atom:
- Protons = 17
- Neutrons = 18
Mass number = 17 + 18 = 35
Isotopes
Definition
Isotopes are atoms of the same element having:
- Same atomic number
- Different mass numbers
They have identical chemical properties because they possess the same number of electrons, but their physical properties differ due to the difference in the number of neutrons in their nuclei.
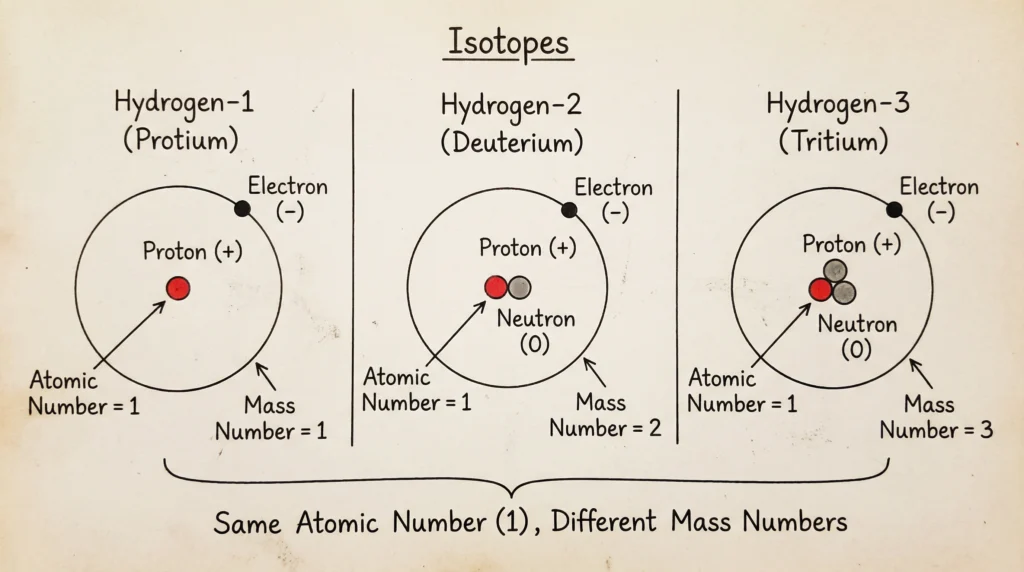
Examples
| Isotope | Protons | Neutrons |
|---|---|---|
| Carbon-12 | 6 | 6 |
| Carbon-14 | 6 | 8 |
Properties of Isotopes
- Same chemical properties
- Different physical properties
Applications
- Carbon dating (C-14)
- Cancer treatment (Co-60)
- Nuclear reactors (U-235)
Real-Life Applications of Atomic Structure
- Medical imaging (X-rays, PET scans)
- Nuclear energy production
- Semiconductor technology
- Radiotherapy in cancer
- Chemical bonding and reactions
Common Mistakes Students Make
- Confusing atomic number with mass number
- Forgetting neutron calculation
- Wrong valency calculation
- Mixing Rutherford and Bohr models
- Incorrect electron distribution
Exam-Oriented Tips
- Memorize Bohr’s postulates
- Practice 2n² formula
- Learn definitions precisely
- Draw neat atomic diagrams
- Revise isotope applications
Worked Examples
Example 1
Find neutrons if atomic number = 11 and mass number = 23.
Solution:
Neutrons = 23 – 11 = 12
Example 2
Find valency of sulfur (atomic number 16).
Electronic configuration: 2,8,6
Valency = 8 – 6 = 2
Practice Questions With Answers
- Define atomic number.
Answer: Number of protons in an atom. - Who proposed Bohr’s atomic model?
Answer: Niels Bohr. - Maximum electrons in M shell?
Answer: 18. - What are isotopes?
Answer: Same atomic number, different mass number. - Valency of magnesium?
Answer: 2.
Quick Revision Summary
The structure of the atom explains how electrons, protons, and neutrons are arranged inside an atom. Starting from Thomson’s model to Rutherford’s nuclear model and finally Bohr’s atomic model, scientists gradually refined our understanding of atomic structure.
Key concepts such as valency, atomic number, mass number, and isotopes are essential for understanding chemical reactions and real-life applications. Mastering this chapter builds a strong foundation for advanced chemistry topics.
A clear understanding of the structure of the atom is not only crucial for exams but also for understanding the material world around us.
For more details, visit Chemistry & Materials
Authoritative References
- BYJU’S & ARPANSA educational resources
- Wikipedia – Atom
- Encyclopaedia Britannica – Atomic structure
FAQs – Structure of the Atom
Q1. Why is atomic number more important than mass number?
Because atomic number identifies the element.
Q2. Why are isotopes chemically similar?
They have same electronic configuration.
Q3. Which model explains atomic stability?
Bohr’s atomic model.
Q4. Can electrons exist between shells?
No, they exist only in fixed energy levels.
Q5. Why do noble gases have zero valency?
Their outer shell is completely filled.
Q6. Are neutrons always necessary for atoms?
Hydrogen-1 has no neutron.
🌐 Connect With Us
👉 Informosio Official – General updates, current affairs, and informative content