What Does “Pure” Really Mean in Chemistry?

In daily life, we often use the word pure to describe things like milk, water, or gold. However, in chemistry, the meaning of purity is very specific and scientific. Chapter 2 – Is Matter Around Us Pure? helps students understand the true nature of matter by explaining mixtures, solutions, methods of separation, and physical and chemical changes in a clear and logical way. When we study Is Matter Around Us Pure?, we begin to realize that many substances we casually call pure are actually mixtures made of more than one component.
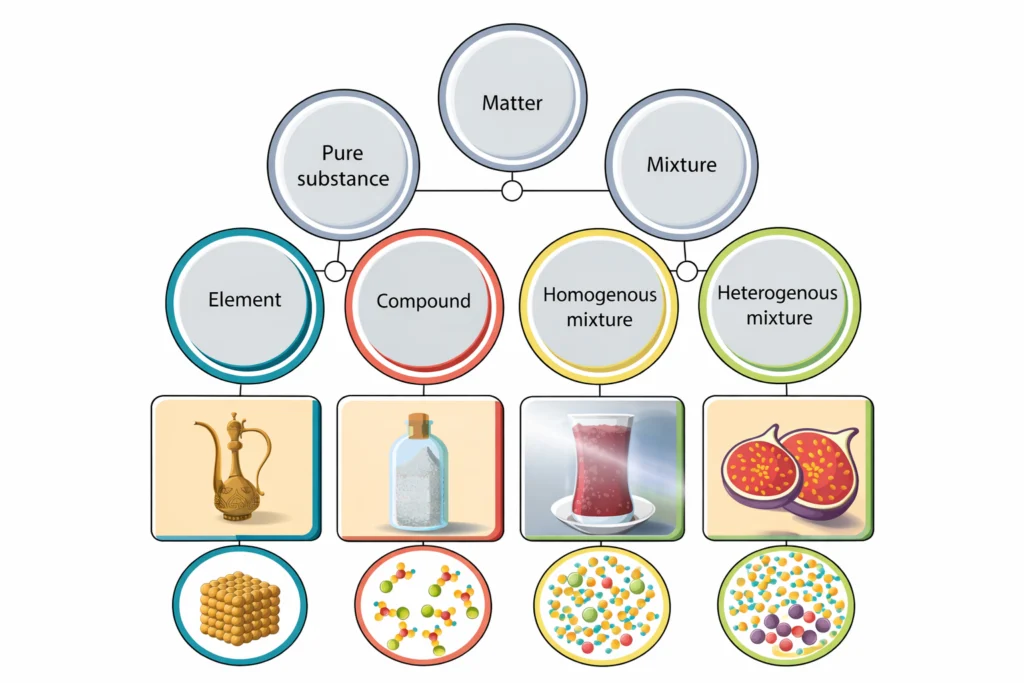
The central idea of Is Matter Around Us Pure? is to distinguish between pure substances and mixtures based on their composition and properties. This chapter explains why elements and compounds are considered pure substances in chemistry, while most materials found in our surroundings are mixtures. By learning Is Matter Around Us Pure?, students gain a scientific understanding of everyday materials such as air, seawater, milk, soil, and alloys.
Is Matter Around Us Pure? also introduces important concepts like solutions, suspensions, and colloids, which help us classify mixtures more accurately. It explains how the components of a mixture can be separated using simple physical methods such as filtration, evaporation, distillation, and chromatography. These ideas form the backbone of practical chemistry and are directly connected to real-life applications like water purification, food processing, and industrial separation techniques.
This chapter forms the foundation of chemistry for Class 9 students and is extremely important for school exams, competitive exams, and higher studies. A strong grasp of Is Matter Around Us Pure? makes it easier to understand advanced topics in chemistry, such as atomic structure, chemical reactions, and states of matter. In this complete guide to Is Matter Around Us Pure?, you will learn each concept step by step with simple definitions, clear explanations, examples, formulas, real-life applications, common mistakes, and exam-oriented tips, making revision easy and effective for students.
What Is Matter?
Matter is anything that has mass and occupies space. Everything around us—air, water, soil, food, metals—is made of matter. In simple terms, if something can be weighed and takes up space, it is called matter. Matter exists in different forms and states, such as solids, liquids, and gases, and it makes up all physical objects in our surroundings.
In chemistry, understanding what matter is becomes the first step toward studying its composition, properties, and behavior. The chapter Is Matter Around Us Pure? builds on this basic idea and helps students explore how matter can be classified based on its purity. By knowing what matter is, students can better understand why substances are grouped as pure substances or mixtures and how their components can be identified and separated using scientific methods.
However, not all matter is the same. Based on composition, matter can be classified into:
- Pure substances
- Mixtures
Understanding this classification is the first step in Chapter 2 – Is Matter Around Us Pure?
Pure Substances in Chemistry

Definition of a Pure Substance
In chemistry, a pure substance is a material that contains only one type of particle and has a fixed composition and fixed properties. It shows uniform chemical characteristics throughout and has definite melting and boiling points. Pure substances cannot be separated into simpler substances by physical methods and include elements and compounds.
Pure substances are of two types:
- Elements
- Compounds
Characteristics of Pure Substances
- Fixed melting and boiling points
- Uniform composition throughout
- Cannot be separated by physical methods
Examples
- Elements: Oxygen, Iron, Gold
- Compounds: Water (H₂O), Sodium chloride (NaCl)
What Is a Mixture?

Definition of a Mixture
A mixture is a substance formed by physically combining two or more substances in any proportion, without any chemical reaction. In a mixture, each component retains its own properties and no new substance is formed.
The composition of a mixture is not fixed, and its components can be separated by simple physical methods such as filtration, evaporation, or distillation. Mixtures are very common in everyday life, for example air, soil, salt solution, and alloys, and they play a key role in the chapter Is Matter Around Us Pure?.
This concept is central to Chapter 2 – Is Matter Around Us Pure? because most materials around us are mixtures.
Key Characteristics of Mixtures
- Variable composition
- Components retain their individual properties
- Can be separated by physical methods
- No fixed melting or boiling point
Examples of Mixtures
- Air (mixture of gases)
- Salt and sand
- Sugar dissolved in water
Types of Mixtures
Mixtures are classified based on uniformity of composition.
Homogeneous Mixtures

Definition
A homogeneous mixture has a uniform composition throughout. The components are not visible separately and are evenly distributed in the mixture.
Such mixtures appear as a single substance and have the same properties in all parts of the mixture. Common examples of homogeneous mixtures include salt solution, sugar solution, and air. Homogeneous mixtures are also known as solutions and are an important concept in the chapter Is Matter Around Us Pure?.
Examples
- Salt solution
- Air
- Vinegar
Homogeneous mixtures are also called solutions.
Heterogeneous Mixtures

Definition
A heterogeneous mixture has a non-uniform composition, and its components can be seen separately. The different substances present in the mixture are not evenly distributed and can be easily identified.
Such mixtures show different properties in different parts of the sample. Common examples of heterogeneous mixtures include sand and water, oil and water, and soil. Understanding heterogeneous mixtures helps students clearly distinguish them from homogeneous mixtures in the chapter Is Matter Around Us Pure?.
Examples
- Sand and water
- Oil and water
- Soil
What Is a Solution?

Definition of a Solution
A solution is a homogeneous mixture of two or more substances. In a solution, the components are uniformly mixed and cannot be seen separately.
A solution consists of two main components: the solute, which is the substance that gets dissolved, and the solvent, which is the substance that dissolves the solute. Common examples of solutions include salt in water, sugar in water, and alcohol in water. Solutions are a key part of the chapter Is Matter Around Us Pure? and help explain the nature of homogeneous mixtures.
Components of a Solution
- Solvent – The substance that dissolves other substances
- Solute – The substance that gets dissolved
Example:
Salt solution
- Solvent: Water
- Solute: Salt
Properties of a Solution
- Uniform composition
- Particles are extremely small (less than 1 nm)
- Do not scatter light
- Stable (particles do not settle down)
Concentration of a Solution
Meaning of Concentration
Concentration refers to the amount of solute present in a given amount of solution. It tells us how much of a substance is dissolved in a solvent and helps compare different solutions based on their strength.
In chemistry, concentration is important because the properties of a solution depend on how concentrated or dilute it is. Concentration can be expressed in different ways, such as mass percentage, volume percentage, or mass by volume, and it is widely used in laboratory work and everyday applications.
Formula: Mass Percentage
Worked Example
If 10 g of salt is dissolved in 90 g of water:
Mass of solution = 100 g
Mass percentage = (10 ÷ 100) × 100 = 10%
Types of Solutions Based on Concentration
- Dilute solution – Small amount of solute
- Concentrated solution – Large amount of solute
- Saturated solution – Cannot dissolve more solute at a given temperature
- Unsaturated solution – Can dissolve more solute
Suspensions
Definition
A suspension is a heterogeneous mixture in which solid particles do not dissolve and settle down on standing. The particles are large enough to be seen with the naked eye and can be separated by filtration.
Suspensions are unstable mixtures and show the Tyndall effect by scattering light. Common examples of suspensions include chalk powder in water and muddy water. Understanding suspensions helps in comparing them with solutions and colloids in the chapter Is Matter Around Us Pure?.

Properties
- Particles are visible
- Scatter light (Tyndall effect)
- Unstable
Example
- Chalk powder in water
Colloids
Definition
A colloid is a mixture in which particles are intermediate in size between solutions and suspensions. The particles are evenly distributed in the medium and are not visible to the naked eye.
Colloids are stable mixtures and show the Tyndall effect by scattering light. The particles do not settle down on standing. Common examples of colloids include milk, fog, blood, and jelly. Colloids are an important concept in the chapter Is Matter Around Us Pure?, as they help explain many natural and everyday substances.

Properties
- Particles not visible to naked eye
- Show Tyndall effect
- Stable
Examples
- Milk
- Fog
- Blood
Difference Between Solution, Colloid, and Suspension
| Property | Solution | Colloid | Suspension |
|---|---|---|---|
| Particle size | Very small | Intermediate | Large |
| Visibility | Not visible | Not visible | Visible |
| Stability | Stable | Stable | Unstable |
| Tyndall effect | No | Yes | Yes |
Separating the Components of a Mixture
Separation techniques are an essential part of Chapter 2 – Is Matter Around Us Pure?
Common Methods of Separation
1. Handpicking
Used when components are large and visible.
Example: Removing stones from rice
2. Winnowing
Used to separate lighter particles from heavier ones using wind.
Example: Separating husk from grains
3. Sieving
Used when particle sizes differ.
Example: Flour and bran
4. Sedimentation and Decantation
Used for insoluble solids in liquids.
Example: Sand and water
5. Filtration
Used to separate insoluble solids from liquids.
Example: Tea leaves from tea
6. Evaporation
Used to separate a dissolved solid from a liquid.
Example: Salt from seawater
7. Distillation
Used to separate liquids with different boiling points.
Example: Alcohol and water
8. Fractional Distillation
Used when boiling points are close.
Example: Separation of petroleum fractions
9. Chromatography
Used to separate colored components.
Example: Dyes in ink
Physical Changes
Definition
A physical change is a change in which no new substance is formed. In a physical change, only the physical properties such as shape, size, or state of a substance change, while its chemical composition remains the same. These changes are usually temporary and reversible, for example melting of ice or dissolving sugar in water.
Characteristics
- Reversible
- Only physical properties change
Examples
- Melting of ice
- Dissolving sugar in water
Chemical Changes
Definition
A chemical change is a change in which new substances are formed. In a chemical change, the chemical composition of the substance changes and the process is usually irreversible. Examples of chemical changes include rusting of iron, burning of paper, and digestion of food.
Characteristics
- Irreversible
- Chemical properties change
Examples
- Rusting of iron
- Burning of paper
Difference Between Physical and Chemical Changes
| Physical Change | Chemical Change |
|---|---|
| No new substance | New substance formed |
| Reversible | Irreversible |
| Temporary | Permanent |
Real-Life Applications
- Purification of drinking water
- Separation of crude oil
- Food processing
- Pharmaceutical industries
- Waste management
Common Mistakes Students Make
- Thinking all clear liquids are pure
- Confusing solutions with compounds
- Writing incomplete separation methods
- Forgetting definitions in exams
Exam-Oriented Tips
- Learn definitions word by word
- Practice numerical problems on concentration
- Draw neat diagrams for separation methods
- Use correct scientific terms
Practice Questions With Answers
- Define a mixture.
A mixture is a physical combination of two or more substances in any proportion. - Name one homogeneous mixture.
Salt solution - Which method is used to separate salt from seawater?
Evaporation - Is rusting a physical or chemical change?
Chemical change
Revision Summary: Chapter 2 – Is Matter Around Us Pure?
In Chapter 2 – Is Matter Around Us Pure?, we learned that most substances around us are mixtures, not pure substances. We studied solutions, colloids, suspensions, and various separation techniques. We also understood the difference between physical and chemical changes, which is crucial for exams.
This chapter builds a strong conceptual base for chemistry and must be revised carefully with definitions, examples, and methods.
You can visit also Chemistry & Materials
Reference Sources For Further Reading
- NCERT Class 9 Science Textbook
- LearnCBSE
- Allen CBSE Notes
- Vedantu
- BYJU’S
- AskIITians
Frequently Asked Questions (FAQs)
Q1. Is air a pure substance?
No, air is a mixture of gases.
Q2. Can mixtures be separated?
Yes, by physical methods.
Q3. Is milk a solution?
No, milk is a colloid.
Q4. What is the solvent in saltwater?
Water
Q5. Is melting of ice a chemical change?
No, it is a physical change.
Q6. Why are compounds pure substances?
They have fixed composition.