Electron Transport Chain (ETC): Complete Guide
The Electron transport chain (ETC) is the final and most crucial step of cellular respiration, where most of the cell’s energy is produced. It occurs inside the mitochondria and plays a central role in converting energy stored in nutrients into ATP, the usable energy currency of the cell. Understanding the ETC is essential for grasping how living organisms sustain life at a molecular level.
At Informosio, In simple terms, the ETC is a series of protein complexes that transfer electrons and generate a proton gradient, ultimately leading to ATP synthesis. This process is highly efficient and tightly regulated, ensuring that cells receive a continuous energy supply.
What is the Electron Transport Chain (ETC)?
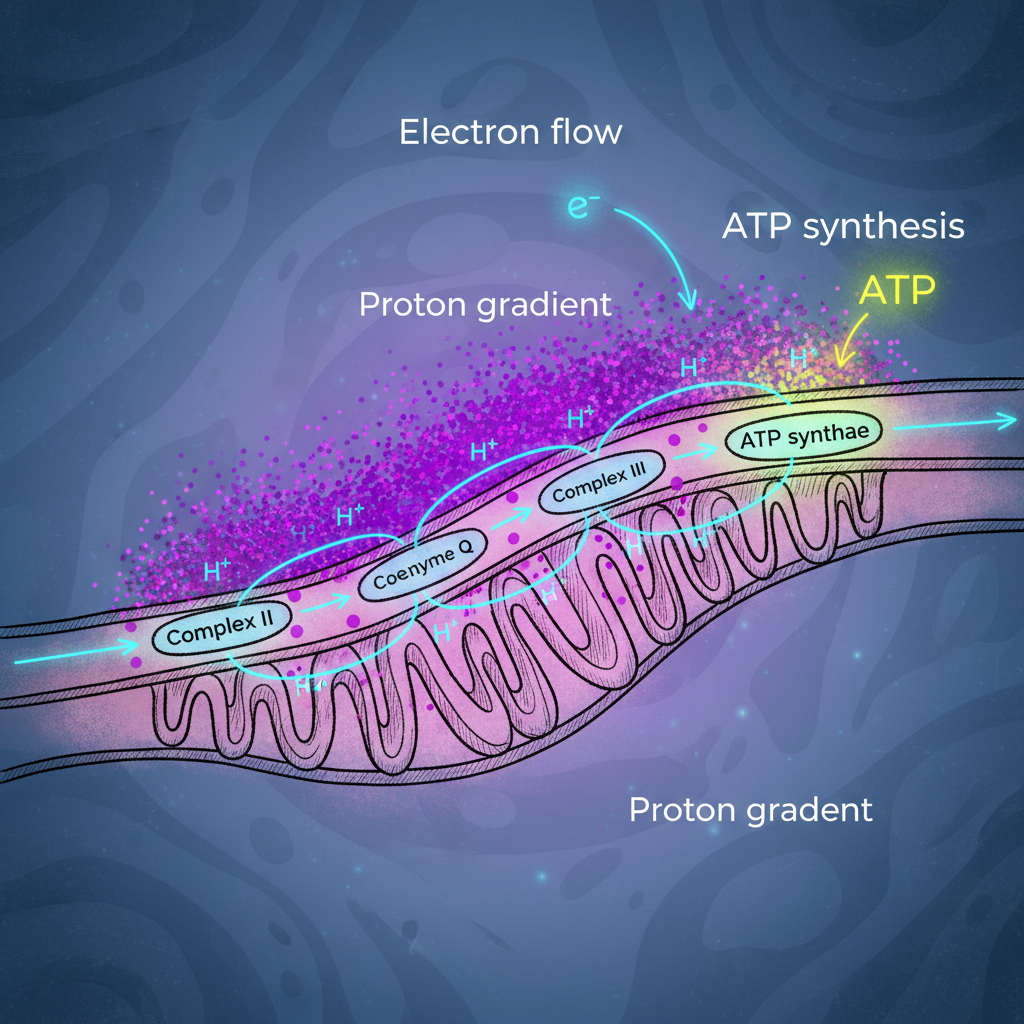
The Electron transport chain (ETC) is a sequence of protein complexes and electron carriers located in the inner mitochondrial membrane. It is responsible for transferring electrons from molecules like NADH and FADH₂ to oxygen, which acts as the final electron acceptor.
This process is coupled with the movement of protons across the membrane, creating a gradient that powers ATP production. Without the ETC, cells would not be able to generate sufficient energy to survive.
The chain consists of four main protein complexes and two mobile carriers. Each component has a specific role in ensuring efficient electron transfer and energy conversion.
Structure of the Electron Transport Chain
The structure of the Electron transport chain (ETC) is highly organized and optimized for energy production. It includes multiple protein complexes embedded in the inner mitochondrial membrane.
Major Components
The ETC consists of the following components:
| Component | Function |
|---|---|
| Complex I | Transfers electrons from NADH |
| Complex II | Transfers electrons from FADH₂ |
| Complex III | Passes electrons to cytochrome c |
| Complex IV | Transfers electrons to oxygen |
| Coenzyme Q | Mobile electron carrier |
| Cytochrome c | Transfers electrons between complexes |
Each of these components works in sequence to ensure smooth electron flow and energy transformation.
How the Electron Transport Chain (ETC) Works
The ETC operates through a series of redox reactions where electrons are passed along the chain. As electrons move, energy is released and used to pump protons across the membrane.
Step-by-Step Process
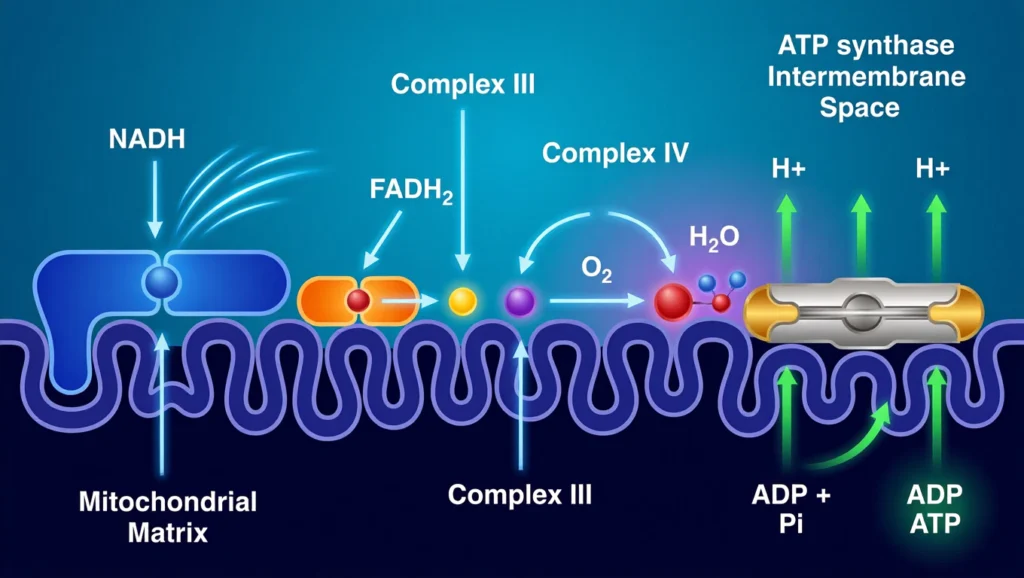
The process begins when NADH and FADH₂ donate electrons to the chain. These electrons move through complexes I to IV, releasing energy at each step. This energy is used to pump protons from the mitochondrial matrix into the intermembrane space.
As protons accumulate, a gradient is formed. This gradient creates potential energy, similar to water stored behind a dam. The protons then flow back into the matrix through ATP synthase, a special enzyme that converts this energy into ATP.
Oxygen plays a critical role as the final electron acceptor. It combines with electrons and protons to form water, ensuring the chain continues to function.
Role of ATP Synthase
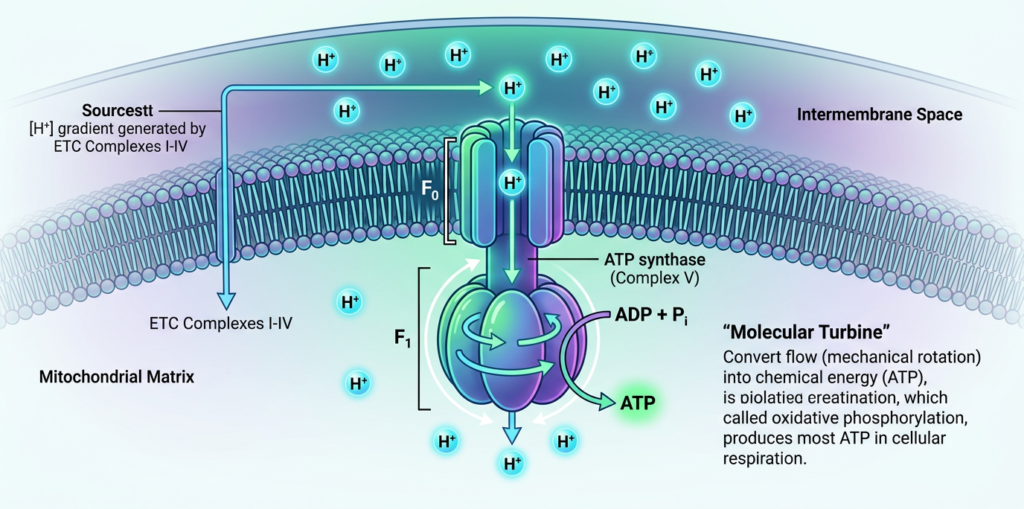
ATP synthase is a key enzyme in the Electron transport chain (ETC). It uses the proton gradient created by the chain to produce ATP. As protons flow through ATP synthase, the enzyme rotates and catalyzes the formation of ATP from ADP and inorganic phosphate. This process is known as oxidative phosphorylation.
ATP synthase is incredibly efficient and produces the majority of ATP generated during cellular respiration.
Importance of the Electron Transport Chain (ETC)
The Electron transport chain (ETC) is essential for life because it produces the bulk of ATP required by cells. Without it, energy production would be severely limited.
This process supports various cellular activities such as muscle contraction, active transport, and biosynthesis. It is especially important in energy-demanding organs like the brain and muscles.
The ETC also helps maintain metabolic balance by regulating electron flow and preventing the buildup of harmful intermediates.
Factors Affecting the Electron Transport Chain
Several factors can influence the efficiency of the Electron transport chain (ETC). These include oxygen availability, temperature, and the presence of inhibitors.
Oxygen is critical because it acts as the final electron acceptor. Without oxygen, the chain stops, and ATP production declines rapidly. This is why oxygen deprivation can be fatal.
Temperature also affects enzyme activity within the chain. Extremely high or low temperatures can disrupt protein structure and reduce efficiency.
Certain chemicals, such as cyanide, can inhibit the ETC by blocking electron transfer. This prevents ATP production and can lead to cell death.
Comparison with Other Stages of Cellular Respiration
The Electron transport chain (ETC) differs significantly from glycolysis and the Krebs cycle in terms of location, output, and efficiency.
| Stage | Location | ATP Produced |
|---|---|---|
| Glycolysis | Cytoplasm | 2 ATP |
| Krebs Cycle | Mitochondrial matrix | 2 ATP |
| Electron Transport Chain (ETC) | Inner mitochondrial membrane | ~28–34 ATP |
This comparison highlights that the ETC is the most productive stage of cellular respiration.
Common Misconceptions
Many people misunderstand the Electron transport chain (ETC) as a simple linear process. In reality, it is a highly dynamic and regulated system. Another misconception is that ATP is directly produced by electron transfer. Instead, ATP is generated through the proton gradient created during the process.
Some also believe oxygen provides energy, but its actual role is to accept electrons and maintain the flow of the chain.
Applications and Real-Life Relevance
The Electron transport chain (ETC) has important applications in medicine and biotechnology. Understanding this process helps researchers develop treatments for metabolic disorders and mitochondrial diseases.
It is also studied in the context of aging and oxidative stress. Damage to the ETC can lead to the production of reactive oxygen species, which contribute to cellular damage.
In biotechnology, manipulating the ETC can improve energy efficiency in microbial systems used for industrial production.
Conclusion
The Electron transport chain (ETC) is the powerhouse of cellular respiration, responsible for generating the majority of ATP in living cells. Its complex yet efficient mechanism ensures that energy stored in nutrients is converted into a usable form.
By understanding the ETC, we gain insight into how life functions at a molecular level. From energy production to its role in health and disease, this process remains one of the most important biological systems.
You may also visit the Biology section.
FAQs
1. What is the main function of the Electron transport chain (ETC)?
The ETC produces ATP by transferring electrons and creating a proton gradient.
2. Where does the ETC occur?
It occurs in the inner mitochondrial membrane.
3. Why is oxygen important in the Electron transport chain (ETC)?
Oxygen acts as the final electron acceptor, allowing the chain to continue functioning.
4. How much ATP does the Electron transport chain produce?
It produces approximately 28–34 ATP molecules per glucose molecule.
5. What happens if the Electron transport chain stops?
ATP production decreases, leading to cellular dysfunction and possible cell death.
6. What molecules donate electrons to the ETC?
NADH and FADH₂ donate electrons to the chain.
7. Is the ETC aerobic or anaerobic?
It is an aerobic process because it requires oxygen.
🌐 Connect With Us
👉 Informosio Tech – Science & Technology news, AI trends, gadgets, and innovations.