A Simple Explanation of Atoms and Molecules
Atoms and Molecules is one of the most important foundational chapters in Chemistry. It explains the basic building blocks of matter, the laws of chemical combination, and the mole concept used for chemical calculations. This chapter helps students understand how atoms combine to form molecules and how substances react in fixed proportions, forming the base for all future Chemistry learning.
Atoms and Molecules: Laws of Chemical Combination
Atoms and Molecules introduces students to the fundamental rules that govern how matter reacts and transforms during chemical reactions. One of the most important parts of Atoms and Molecules is the study of the laws of chemical combination, which provide scientific proof that matter follows fixed and predictable patterns.
The laws of chemical combination explain how substances react and combine with each other during chemical reactions. These laws were established through repeated experimental observations.
In Atoms and Molecules, these laws help students understand that chemical reactions are not random. Instead, atoms rearrange in a definite manner while obeying strict mass relationships. The two main laws discussed in Atoms and Molecules are the Law of Conservation of Mass and the Law of Constant Proportions.
Through Atoms and Molecules, students learn that mass remains conserved in every chemical reaction and that compounds always have a fixed composition by mass. This knowledge forms the foundation for balancing chemical equations, solving numericals, and understanding advanced Chemistry topics.
Overall, Atoms and Molecules uses the laws of chemical combination to strengthen logical thinking, experimental understanding, and exam-oriented problem-solving skills, making it a core chapter in the Chemistry syllabus.
Law of Conservation of Mass
Statement
Mass is neither created nor destroyed in a chemical reaction. This statement means that during any chemical reaction, the total mass of the reactants before the reaction is always equal to the total mass of the products formed after the reaction. Atoms are only rearranged, not created or destroyed.
Explanation
In any chemical reaction, the total mass of the reactants before the reaction is equal to the total mass of the products after the reaction.
Atoms are only rearranged during a reaction; they are not created or destroyed.
Example
Consider the reaction:
2H₂ + O₂ → 2H₂OIf:
- Hydrogen = 4 g
- Oxygen = 32 g
Total mass of reactants = 36 g
Total mass of products (water) = 36 g
This confirms the law of conservation of mass.
Importance
- Helps in balancing chemical equations
- Forms the basis of stoichiometric calculations
- Confirms the particulate nature of matter
Law of Constant Proportions
Statement
A pure chemical compound always contains the same elements combined in the same fixed proportion by mass, regardless of its source. This law states that the composition of a chemical compound is constant and does not depend on how the compound is prepared or from where it is obtained. Whether a compound is formed in a laboratory, extracted from natural sources, or produced by a chemical reaction, the ratio of the masses of its constituent elements remains unchanged. This law helps distinguish pure compounds from mixtures and confirms that chemical substances have a definite and uniform composition.
Explanation
The composition of a compound does not change. For example, water always contains hydrogen and oxygen in the mass ratio 1:8.
Example
In water (H₂O):
- Hydrogen = 1 part by mass
- Oxygen = 8 parts by mass
This ratio remains constant whether water is obtained from a river, rain, or laboratory preparation.
Significance
- Confirms that compounds have fixed composition
- Distinguishes compounds from mixtures
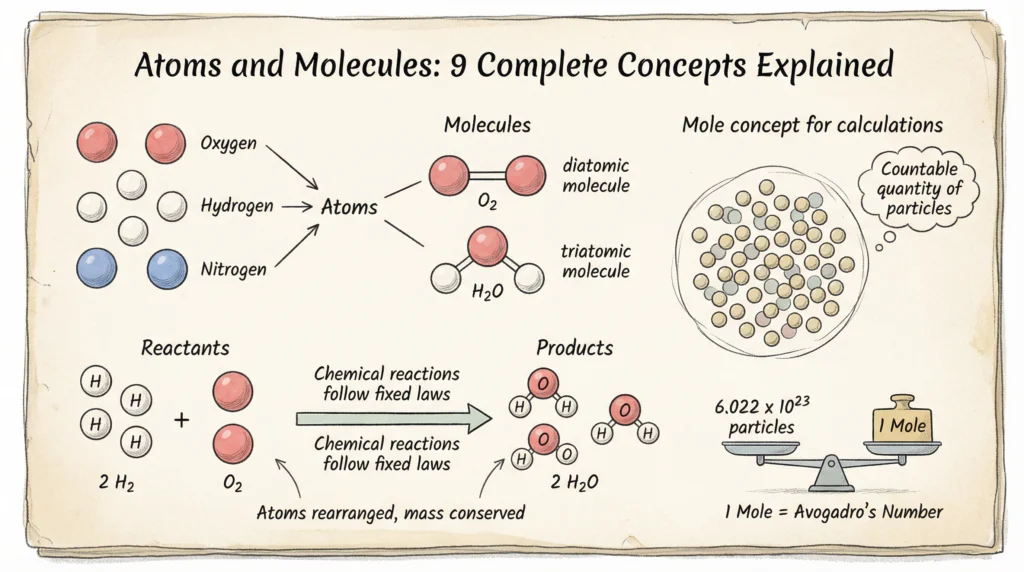
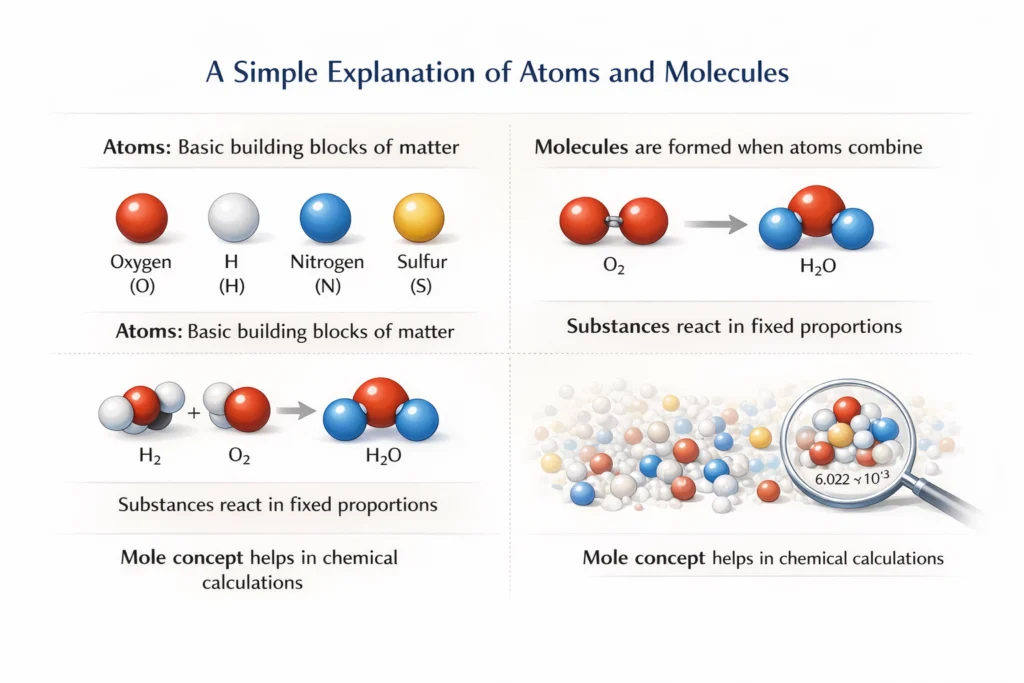
What Is an Atom?
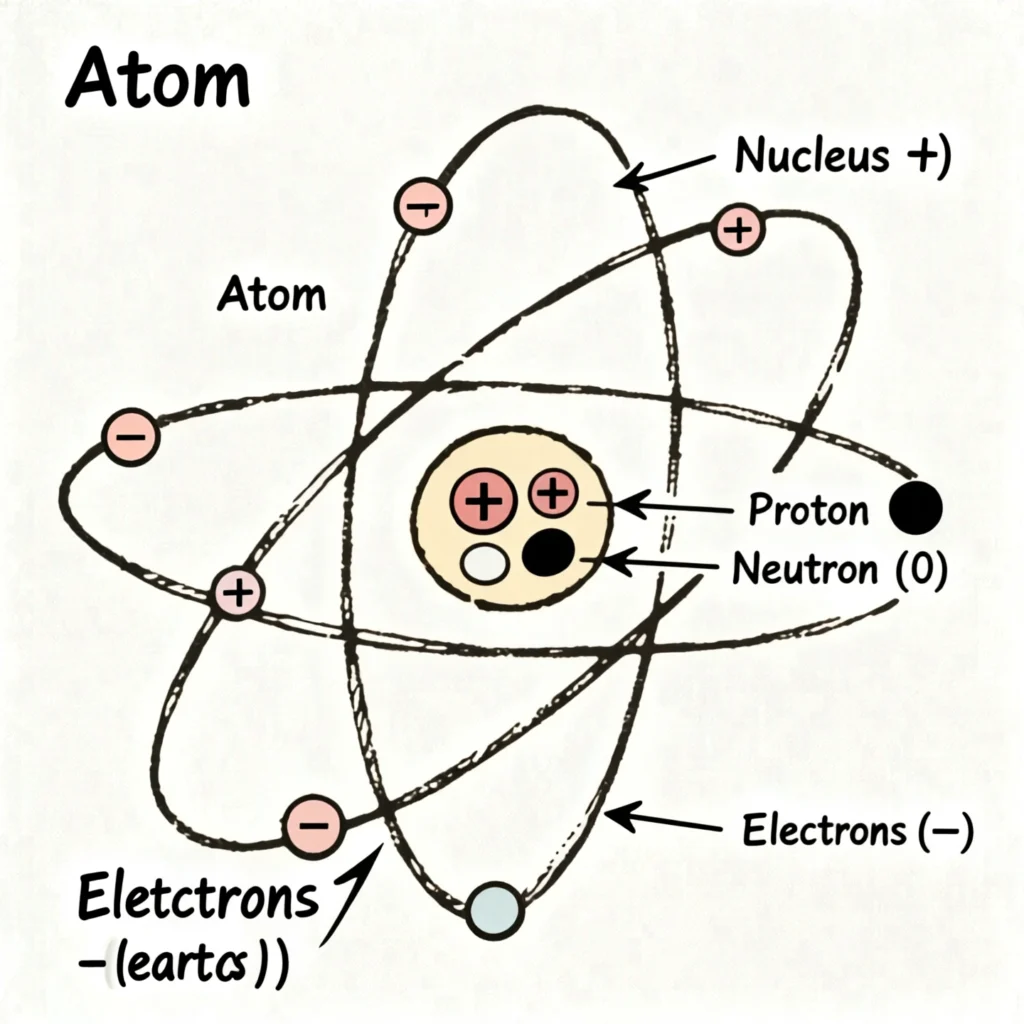
An atom is the smallest particle of an element that retains the chemical properties of that element. Atoms are extremely small in size and cannot be seen with the naked eye. The structure of the atom consists of a central nucleus containing protons and neutrons, with electrons revolving around it. Atoms participate in chemical reactions and combine with other atoms to form molecules. They usually do not exist independently and are the basic building blocks of all matter.
Key Characteristics of Atoms
- Extremely small in size
- Participate in chemical reactions
- Cannot be divided by chemical methods
- Usually do not exist independently
Examples
- Hydrogen atom (H)
- Oxygen atom (O)
- Carbon atom (C)
Atoms combine to form molecules during chemical reactions.
Symbols of Elements
To represent atoms, scientists use symbols.
Rules for Writing Symbols
- One or two letters
- First letter is capital
- Second letter (if any) is lowercase
Examples
- Hydrogen → H
- Oxygen → O
- Sodium → Na
- Iron → Fe
Symbols provide a universal language for Chemistry.

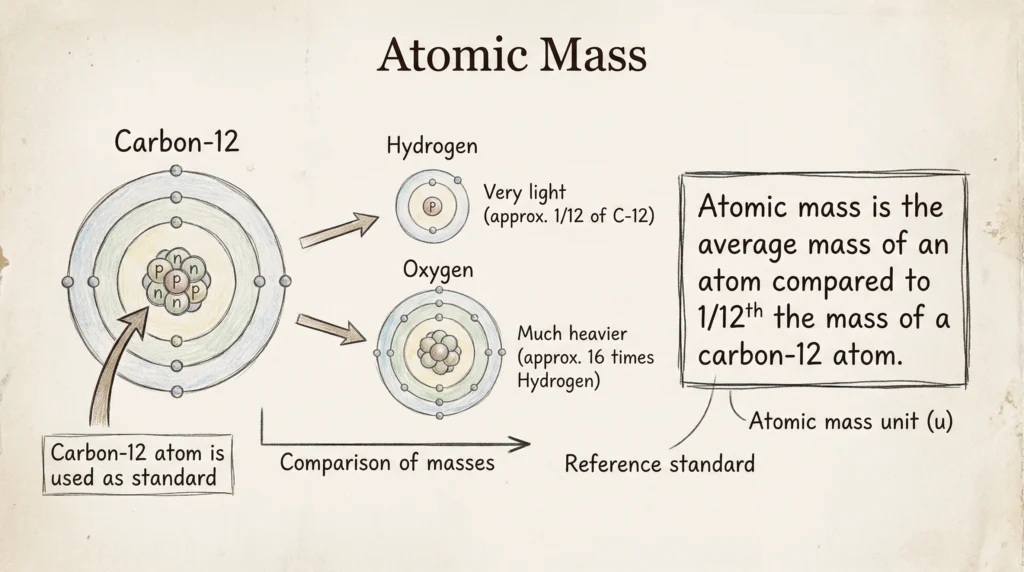
Atomic Mass
Definition
Atomic mass is the average mass of an atom of an element compared to 1/12th the mass of a carbon-12 atom. It is expressed in atomic mass units (u) and represents the relative mass of atoms of different elements. Atomic mass helps in comparing elements and is used to calculate molecular mass and molar mass in Chemistry.
Unit
Atomic mass unit (u)
Examples
- Hydrogen = 1 u
- Carbon = 12 u
- Oxygen = 16 u
Atomic mass helps in calculating molecular mass and mole-based quantities.
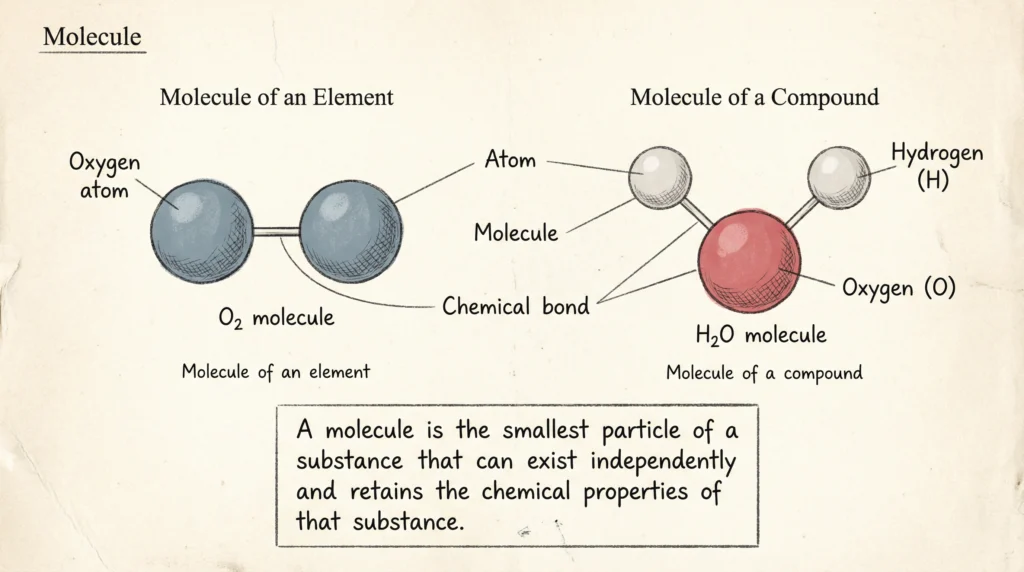
What Is a Molecule?
A molecule is the smallest particle of a substance that can exist independently and retains the chemical properties of that substance.
Molecules are formed when two or more atoms combine chemically in a fixed ratio. They may consist of atoms of the same element, such as oxygen (O₂), or atoms of different elements, such as water (H₂O).
Molecules are formed when atoms combine chemically.
Types of Molecules
Molecules of Elements
Formed by atoms of the same element.
Examples
- Oxygen (O₂)
- Nitrogen (N₂)
- Hydrogen (H₂)
Molecules of Compounds
Formed by atoms of different elements.
Examples
- Water (H₂O)
- Carbon dioxide (CO₂)
- Ammonia (NH₃)
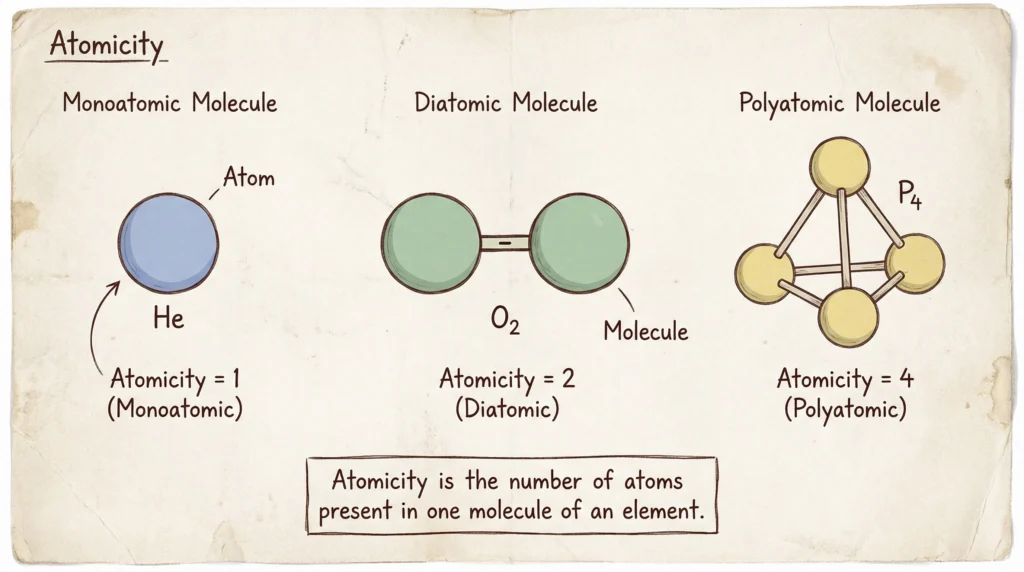
Atomicity
Definition
Atomicity is the number of atoms present in one molecule of an element. It helps in classifying elements as monoatomic, diatomic, or polyatomic based on the number of atoms in their molecules. For example, helium has atomicity 1, oxygen has atomicity 2, and phosphorus has atomicity 4.
Examples
- Phosphorus (P₄): 4 (Polyatomic)
- Helium (He): 1 (Monoatomic)
- Oxygen (O₂): 2 (Diatomic)
- Ozone (O₃): 3 (Triatomic)
Chemical Formula
A chemical formula represents a compound using symbols and subscripts to show the number of atoms of each element.
Examples
- Water → H₂O
- Carbon dioxide → CO₂
- Calcium chloride → CaCl₂
Chemical formulae show the composition of substances clearly and concisely.
Molecular Mass
Definition
Molecular mass is the sum of atomic masses of all atoms present in a molecule. It is calculated by adding the atomic masses of each element in the molecular formula and is expressed in atomic mass units (u). Molecular mass is useful for determining molar mass and for solving numerical problems in Chemistry.
Formula
Molecular Mass = Sum of atomic massesExample
Molecular mass of water (H₂O):
- Hydrogen = 1 × 2 = 2 u
- Oxygen = 16 × 1 = 16 u
Total = 18 u
Atoms and Molecules: The Mole Concept
The mole concept is one of the most important parts of the chapter Atoms and Molecules. It helps in counting atoms and molecules using mass.
What Is a Mole?
A mole is the amount of substance that contains the same number of particles as there are atoms in 12 g of carbon-12. This fixed number of particles is known as Avogadro’s number (6.022 × 10²³). The mole is a standard unit used to measure atoms, molecules, or ions and is essential for performing chemical calculations.
Avogadro’s Number
1 mole = 6.022 × 10²³ particlesParticles may be atoms, molecules, or ions.
Molar Mass
Definition
Molar mass is the mass of one mole of a substance. It is expressed in grams per mole (g/mol) and is numerically equal to the molecular mass of the substance. Molar mass is used to convert between mass, moles, and number of particles in chemical calculations.
Unit
g/mol
Examples
- Molar mass of O₂ = 32 g/mol
- Molar mass of H₂O = 18 g/mol
Relationship Between Mole, Mass, and Number of Particles
Important Formulae
Number of moles (n) = Given mass / Molar mass
Number of particles = n × Avogadro’s numberWorked Numerical Examples
Example 1
Find the number of moles in 36 g of water.
Solution
- Molar mass of H₂O = 18 g/mol
- Moles = 36 ÷ 18 = 2 mol
Example 2
Find the number of molecules in 1 mole of oxygen gas.
Solution
- 1 mole = 6.022 × 10²³ molecules
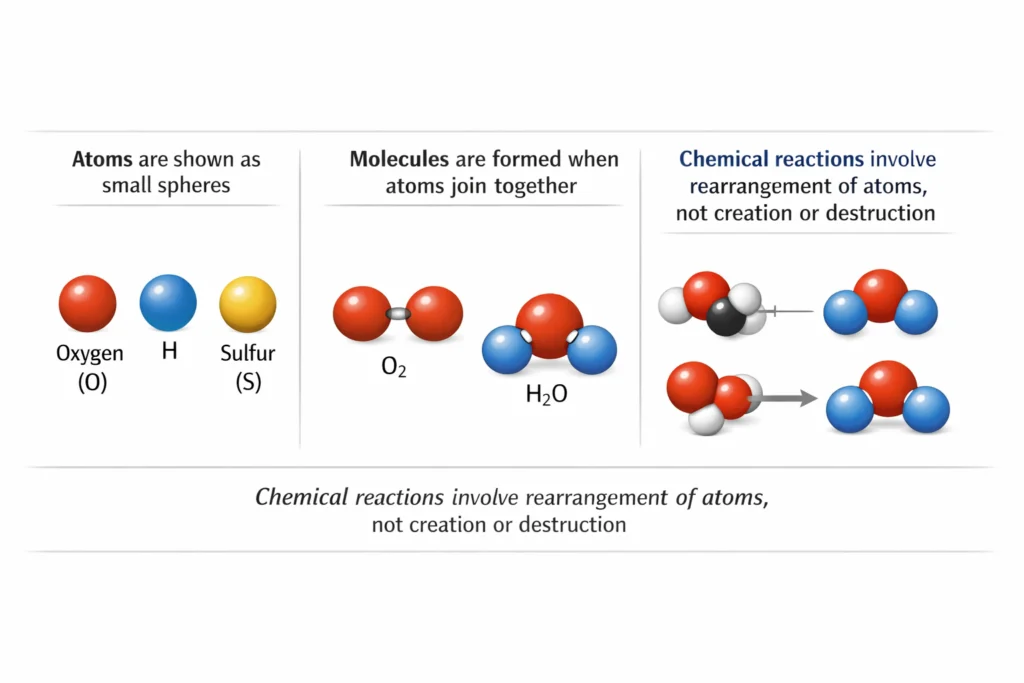
Explaining Diagrams
- Atoms are shown as small spheres
- Molecules are shown as two or more spheres joined together
- Chemical reactions show rearrangement of atoms, not creation or destruction
Understanding diagrams helps visualize particle behavior.
Real-Life Applications of Atoms and Molecules
- Medicines are measured in moles
- Fertilizers are calculated using molar mass
- Industrial chemicals follow laws of chemical combination
- Cooking involves chemical reactions governed by mass conservation
Common Mistakes Students Make
- Confusing atomic mass with molar mass
- Forgetting Avogadro’s number
- Incorrect unit conversions
- Writing wrong chemical formulae
- Ignoring subscripts in molecules
Exam-Oriented Tips
- Always write units in numerical problems
- Memorize key formulae
- Balance equations carefully
- Show full steps in calculations
- Learn definitions word by word
Practice Questions
Question 1
Define the law of conservation of mass.
Answer
Mass is neither created nor destroyed in a chemical reaction.
Question 2
Calculate the molecular mass of CO₂.
Answer
- Carbon = 12 u
- Oxygen = 16 × 2 = 32 u
Total = 44 u
Question 3
How many moles are present in 11 g of carbon dioxide?
Answer
- Molar mass = 44 g/mol
- Moles = 11 ÷ 44 = 0.25 mol
Quick Revision Summary
The chapter Atoms and Molecules explains the fundamental structure of matter and the rules that govern chemical reactions. By understanding the laws of chemical combination, the nature of atoms and molecules, and the mole concept, students gain the ability to analyze and calculate chemical changes accurately.
Mastering Atoms and Molecules builds a strong foundation for future Chemistry topics and ensures confidence in numerical problem-solving and conceptual understanding. Regular practice, clear definitions, and careful calculations are the keys to success in this chapter.
For more details, visit Chemistry & Materials
Frequently Asked Questions (FAQs)
1. Why is the chapter Atoms and Molecules important?
It forms the foundation for all chemical calculations and reactions.
2. What is the difference between atom and molecule?
An atom is the smallest unit of an element, while a molecule is formed when atoms combine.
3. What is Avogadro’s number used for?
It helps count atoms and molecules using moles.
4. Can atoms exist independently?
Most atoms cannot exist independently except noble gases.
5. What is molar mass?
It is the mass of one mole of a substance.
6. Why is the mole concept important in exams?
It is essential for solving numerical problems.
🌐 Connect With Us
👉 Informosio Official – General updates, current affairs, and informative content